News

09 October 2025
2025 Lab Coat Ceremony Welcomes Graduate Students To Their Thesis Labs
An annual tradition marks the start of scientific discovery for the 2024-2025 class of Stowers Graduate School students.
Read Article
News
Though presently incurable, research from Stowers Associate Investigator Randal Halfmann, Ph.D., is helping improve our understanding of how the ALS amyloid protein that forms aggregates in the brain first starts. Recently, the team made an unexpected, exciting discovery that offers hope for the future.
By Rachel Scanza, Ph.D.
Amyotrophic lateral sclerosis (ALS), a neurodegenerative disease affecting 1 in 400 individuals, is characterized by a progressive loss of motor neurons that impact walking, eating, swallowing, and eventually breathing. Though presently incurable, research from Stowers Associate Investigator Randal Halfmann, Ph.D., is helping improve our understanding of how the ALS amyloid protein that forms aggregates in the brain first starts. Recently, the team made an unexpected, exciting discovery that offers hope for the future.
The Halfmann Lab’s research, which is focused on aging and age-related diseases, explores first principles into how amyloid-associated proteins initially spark the aggregation process found in neurodegenerative conditions like Alzheimer’s, Huntington’s, and ALS.
“I am interested in what is happening at the protein level for the processes of aging that manifest as age-associated diseases like ALS and Alzheimer’s,” said Halfmann. “We want to understand the moment these diseases start—the very first thing that happens to a particular protein that initiates the chain reaction toward aggregation.”
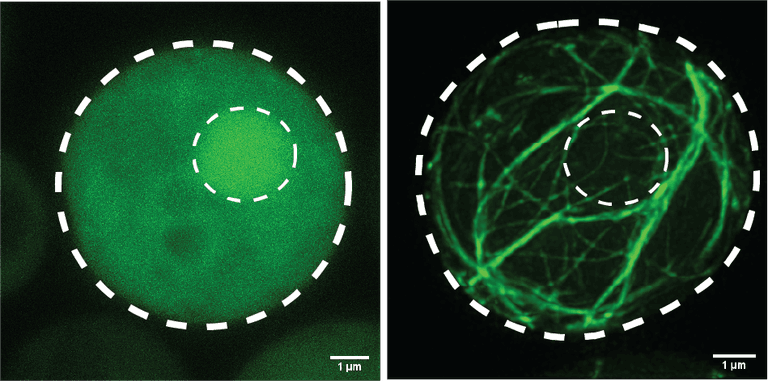
Two yeast cells expressing the pathogenic fragment of the ALS-causing protein, TDP-43. The Halfmann Lab is studying the intrinsic properties of the protein that cause it to transition from the left image (soluble, healthy) to the right image (amyloid, diseased).
Some proteins essential for cells to function have the potential for a random change of shape, altering their behavior from beneficial to toxic. Uncovering that crucial moment governing that change may reveal a way to prevent its occurrence.
“A sequence of ‘letters’ make up a protein and we found something new,” said Halfmann. “We found ‘the letter’ in the ALS protein’s sequence that determines whether or not it aggregates and identified other proteins that can control a switch in that letter. And it is showing us a possible therapeutic path forward.”
Those letters are called amino acids and are the building blocks of proteins. Technology developed at the Stowers Institute gives Halfmann and his team the ability to provide tailored protein production instructions to single-celled yeast to then measure the protein’s ability to aggregate. Why yeast? They do exactly what you tell them to, are easy to analyze, and can capture a spontaneous fluctuation in protein shape that might get lost in the more complicated environment of neurons.
“There are genetic reasons for developing ALS but there is also spontaneity, or things we don’t understand,” said Halfmann. “When we use our yeast cultures, we can eliminate all the genetic factors and get a sense of what those missing spontaneous factors are.”
Moving forward, the team is testing their hypotheses in more complex systems, from yeast, to neurons in a dish, to organoids, to mice. But additionally, they are discovering that very different neurodegenerative diseases are illuminating common themes, another step toward a comprehensive understanding and the hope for effective treatments and a cure.
“Something that has emerged recently is that the protein accumulating as aggregates in nearly all ALS patients is doing the same thing in about half of all patients with Alzheimer’s-like dementia,” said Halfmann. “As we learn more about ALS, we are going to learn more about Alzheimer’s and vice versa. Studying this broad category as a whole may allow us to make progress on all age-associated diseases.”
News

09 October 2025
An annual tradition marks the start of scientific discovery for the 2024-2025 class of Stowers Graduate School students.
Read Article
In The News

17 September 2025
From New Scientist, Inflammation is a vital part of the immune response, but it seems that the system can sometimes go awry, resulting in chronic inflammation that has been linked to conditions such as cancer.
Read Article
News

29 July 2025
"Most of my best ideas—and opportunities—came from conversations with other scientists. Share data, workshop problems, collaborate. It’s the single most valuable habit I’ve cultivated."
Read Article